
Coronavirus
Technology Solutions
February 17, 2021
Markets for Air
Filters,
Controls and
Masks Changing
Rapidly
Experts Call on
Biden and OSHA
to Require Masks
With the New
Level 2 80%
Efficiency
Rating and for a
CDC Consumer
Mandate as Well
New ASTM
Standards to be
Discussed on
March 2 INDA
Webinar
Freudenberg
Receives FDA
Clearance for
Surgical Masks
Lydall Urges
Consumers to Buy
ASTM-Certified
and Buy
American-Made
Handanhy
Supplies Melt
Blown Media and
Masks Worldwide
Mann + Hummel
Room Air
Purifier
Provides
Effective
Protection From
Viruses in
Classrooms
Capture Virus
and Dust at the
Source - Brake
Filter Example
It will Take
Years to
Vanquish
COVID-19
________________________________________________________________________
Markets for Air
Filters,
Controls and
Masks Changing
Rapidly
New ASTM mask
standards
published this
week will very
likely result in
the demise of a
$15
billion
U.S. market for
cloth masks and
help create an
even larger
market for masks
with efficient
filter media.
The pandemic is
changing the
entire market
for air filters,
monitoring
/controls as
well as masks.
To date
the impact has
been a
nationalistic
trend but long
term the result
will be the
opposite.
For example the
U.S. will be
best served by
ramping up
efficient masks
capacity now and
then subsidizing
exports in
future years
when the
domestic needs
subside.
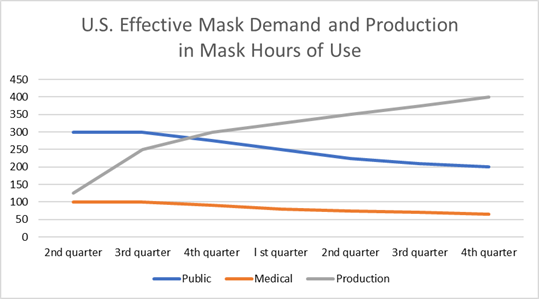
The Chinese
economy is
booming due to
success in
minimizing the
pandemic impact.
Some of the
other countries
of the world
will not achieve
herd immunity
for years and
will be
subjected to
more deadly
variants of the
virus. Therefore
the
markets
for filters,
monitoring/controls,
and masks will
continue to be
significantly
impacted.
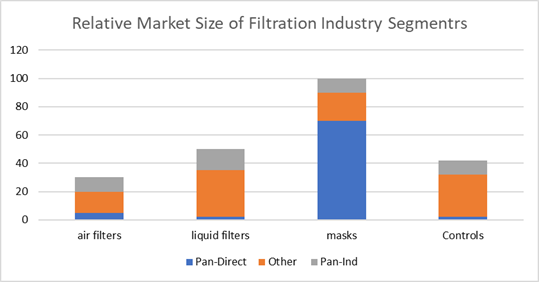
The near term
opportunity for
efficient masks
will greatly
exceed any other
filter market.
Suppliers
need to
carefully assess
all the impacts
and to be
proactive.
Many of the
suppliers are
international.
They have a
unique
opportunity to
help shape the
world markets.
The
McIlvaine
Company with its
extensive
international
research and
analysis
provides
strategic
support for
suppliers in
this fast moving
market.
For more
information on
the strategic
support services
contact Bob
McIlvaine at 847
226 2391 or
rmcilvaine@mcilvainecompany.com.
Experts Call on
Biden and OSHA
to Require Masks
With the New
Level 2 80%
Efficiency
Rating and for a
CDC Consumer
Mandate as Well
Several members
of President
Joe Biden's former coronavirus advisory
board are urging
his
administration
to more widely
recommend and
mandate the use
of N95
masks,
citing a
"pressing and
urgent need for
action" driven
by the threat of
new coronavirus
variants.
In a memo to
Biden's top
coronavirus
advisers
obtained by CNN,
a dozen health
and safety
experts --
including four
members of
Biden's former
advisory board
-- called on the
US Centers for
Disease Control
and Prevention
and the
Occupational
Safety and
Health
Administration
(OSHA) to
"recommend and
require the use
of respiratory
protection, such
as N95 FFRs
(filtering
facepiece
respirators), to
protect all
workers at high
risk of exposure
and infection."
They also urged
the CDC to adopt
the first
national
consumer mask
standard and
urged the
administration
to "coordinate a
national effort"
to distribute
National
Institute for
Occupational
Safety and
Health-certified
respirators and
ASTM barrier
face coverings
to workers in
need and use the
Defense
Production Act
to ramp up mask
production.
The memo, which
is addressed to
Biden's
coronavirus czar
Jeff Zients, CDC
Director Dr.
Rochelle
Walensky and Dr.
Anthony Fauci,
came just days
after the CDC
updated its
guidance on face
masks. The new
guidance for the
first time
heralded the
benefits of
double-masking
but maintained a
recommendation
against the use
of N95 masks "in
non-healthcare
settings,"
citing a
shortage of
N95s.
But the memo's
signatories --
including former
Biden advisers
Rick Bright, Dr.
Celine Gounder,
Dr. David
Michaels and
Michael
Osterholm -- say
there are now
millions of N95
masks "available
and sitting in
warehouses," and
are urging the
administration
to go further.
"While COVID-19
infections and
deaths have
started to
decline in
recent weeks,
they remain at a
very high level
and, unless
strengthened
precautionary
measures are
implemented, the
new variants
will likely
bring an
explosion in new
infections," the
experts write in
the memo.
"Action is
needed to better
protect workers
and the public
against
inhalation
exposure to the
virus."
Bright, Gounder,
Michaels and
Osterholm were
part of Biden's
16-member
coronavirus
advisory board,
which Biden
assembled during
the transition
to advise him on
the pandemic and
help him craft a
response
strategy. The
board was
dissolved once
Biden was
inaugurated and
its three
co-chairs joined
the
administration.
Bright
previously
headed the
Department of
Health and Human
Services'
biotechnology
research arm
before becoming
a whistleblower
in the early
months of the
Trump
administration's
coronavirus
response.
Michaels is an
epidemiologist
who headed OSHA
in the Obama
administration.
Gounder and
Michaels are
both infectious
disease experts.
The former Biden
advisers were
joined in
writing the memo
by leading
public health
and occupational
safety experts,
including Lisa
Brosseau, Dr.
Lynn Goldman,
Dr. Yoshihiro
Kawaoka, Linsey
Marr, Dr. Donald
Milton, Kimberly
Prather, Dr.
Robert Schooley
and Peg
Seminario.
In a statement
responding to
the letter, the
CDC touted the
effectiveness of
"properly worn
... well-fitting
cloth masks" and
pointed to a CDC
study showing
that "exposure
to aerosol
particles was
reduced by more
than 95%" when
double-masking
or wearing a
tightly-fitting
surgical mask.
"For reasons
supported by
science,
comfort, costs,
and
practicality,
CDC does not
recommend the
use of N95
respirators for
protection
against COVID-19
by the general
public," the CDC
said in a
statement,
though it did
not address the
question of
recommending N95
masks to
high-risk
workers.
The memo follows
a similar letter
from some
Democratic
lawmakers sent
on February 1,
which called on
Biden to
increase the
supply and
availability of
higher quality
masks and
encourage the
education of the
public on which
masks are most
effective.
The latest memo
urges the
administration
to follow the
example of
several European
countries like
Germany and
France, which
have mandated
the use of
N95-style masks
and
higher-quality
masks in
workplaces and
public places.
The authors also
called on the
CDC to adopt the
first national
consumer mask
standard to
protect American
workers and
general public
established by
ASTM, an
international
technical
standards
organization.
That standard
outlines minimum
fit, design,
performance and
testing
requirements for
consumer face
masks and will
soon allow the
public to be
able to choose
between two
levels of mask
protection seen
on package
labeling.
The letter also
asks OSHA to
utilize the ASTM
standard and
offer
high-performing
face masks with
at least 80%
filtration
efficacy to
non-health care
workers at lower
exposure risks.
While the memo's
authors praised
the
administration's
coronavirus
response plan,
the memo is one
of the first
signs of outside
pressure urging
the White House
to do more amid
the threat of
new, more
transmissible
coronavirus
variants.
In the memo, the
experts urge the
administration
to rescind
recommendations
advising
healthcare
workers not
involved in
direct patient
care and
high-risk
workers like
those in meat
plants to only
wear face
coverings or
surgical masks
-- rather than
N95s.
In a nod to
Biden's focus on
equity in his
coronavirus
response, the
authors also
noted that the
risks of
transmission are
especially acute
for people of
color.
"The failure to
address
inhalation
exposure to
SARS-CoV-2
continues to put
workers and the
public at
serious risk of
infection,
particularly
people of color,
many of whom
work on the
front lines in
essential jobs
and have
suffered -- and
continue to
suffer -- the
greatest impacts
of the COVID-19
pandemic," the
memo says.
New ASTM
Standards to be
Discussed on
March 2 INDA
Webinar
INDA, the
Association of
the Nonwoven
Fabrics
Industry,
announced a
webinar date of
Tuesday, March
2, at 11 a.m.
EST to discuss
the new ASTM
F3502-21
Standard
Specification
for Barrier Face
Coverings
launched by ASTM
on February 15.
This standard
establishes for
the first time a
set of test
methods that
evaluate the
filter, fit and
leakage
performance of
barrier face
coverings,
commonly
referred to as
“face masks”
worn by the
general public,
and not to be
confused with
respirators nor
medical or
surgical masks.
The webinar will
be presented by
respiratory
expert, Jeff
Stull, vice
chair of the
ASTM Committee
that wrote the
standard, and
Dave Rousse,
INDA president.
It will provide
a detailed
review of the
new ASTM
standard and the
test methods it
entails and the
impact on the
entire supply
chain of
facemask
production.
Details and
registration are
available here:
www.inda.org/inda-webinars
The purpose of
guidance for the
general public
to wear face
masks is to
control the
spread of
viruses. Face
masks made to
this new ASTM
standard will
also provide a
degree of
particulate
filtration to
reduce the
amount of
inhaled
particulate
matter.
The goal of the
standard is to
assist consumers
in making
informed
decisions about
face masks given
the vast array
of products
currently for
sale, including
various patterns
promoted for
homemade
manufacture
using common
textile
materials. Prior
to the ASTM
standard, no
standard test
method existed
which allowed
comparisons
among different
products nor
were there any
minimum
performance
requirements.
This new
standard
provides these
performance
requirements as
well as a set of
specifications,
guidelines and
expectations for
face mask
manufacturers
and media
suppliers.
“We approached
NIOSH last year
on developing a
general public
face mask
standard that
could use
nonwoven
materials beyond
meltblown that
still deliver an
effective level
of filtration,
as there was so
much demand for
the N95
respirators and
masks once the
Asian supply
chain was cut
off,” said Dave
Rousse, INDA
president. “We
were delighted
to get a
positive
response from
Jon Szalajda,
NIOSH deputy
director,
National
Personal
Protective
Technology
Laboratory, who
is also the
chair of the
ASTM Committee
dealing with
standards in
this area.”
“This was a
worthwhile
project that we
worked through
the ASTM process
in record time,”
Szalajda said.
“It should
provide an
important
benefit in the
fight against
COVID-19 spread
by reducing
consumer
confusion about
what works and
what does not
and assisting
manufacturers in
making effective
products.”
“The development
of this standard
has been
followed by the
Occupational
Safety & Health
Administration
(OSHA),” Rousse
said, “as it
considers the
January 21
Presidential
Executive Order
on whether any
emergency
temporary
standards with
respect to masks
in the workplace
are necessary to
reduce the risk
of COVID-19
spread, and if
so, to issue
them by March
15. This would
have a
significant and
rapid impact on
the facemask
industry. In our
March 2 webinar,
we will review
all of these
developments and
provide needed
information and
guidance to the
suppliers,
converters and
marketers in
this important
sector.”
This webinar on
March 2 will be
in addition to
the INDA Webinar
Series already
scheduled for
March 16, March
25 and April 6.
For information
on the full
webinar series,
visit
www.inda.org/inda-webinars.
Freudenberg
Receives FDA
Clearance for
Surgical Masks
ASTM Level 3
surgical masks
manufactured by
Freudenberg
Performance
Materials
recently
received 510(k)
clearance from
the US Food and
Drug
Administration
(FDA). The
surgical masks
are intended for
use by
healthcare
personnel to
protect both the
patient and them
from transfer of
microorganisms,
body fluids and
particulate
material.
The face masks
are intended for
use in infection
control
practices to
reduce the
potential
exposure to
blood and body
fluids.
For the U.S.
market,
Freudenberg
Performance
Materials is now
providing FDA
cleared surgical
masks meeting
the Level 3
standard of the
American Society
of Testing and
Materials
(ASTM). The ASTM
Level 3 surgical
mask is for use
in conditions
where there is a
high risk of
fluid and spray
of aerosol
transmission,
such as
operating
procedures. The
masks are a
single use,
disposable
device provided
non-sterile.
Surgical masks
by Freudenberg
were tested for
performance in
five areas:
fluid
resistance,
differential
pressure,
particulate
efficiency,
bacterial
filtration
efficiency and
flammability.
Upon completion
of testing, the
surgical masks
consistently met
ASTM Level 3
criteria in all
five performance
test areas.
Bacterial and
particle
filtration
efficiency test
results were
greater than
99%.
“This was a
great
accomplishment
for our team. It
is a milestone
that supports
our commitment
to continue
manufacturing
local. Meeting
the ASTM Level 3
standard helps
us in our goal
to not only keep
our healthcare
workers safe,
but also ensure
they have a
steady, reliable
PPE supply so
the shortages
that happened at
the beginning of
the pandemic
don’t happen
again,” says
Alicen Pittenger,
Head of Sales –
North America –
Global
Healthcare
Division,
Freudenberg.
The Durham, NC
site of
Freudenberg
Performance
Materials began
mask production
in 2020 in
response to the
COVID-19
pandemic.
Working closely
with local
partners,
Freudenberg is
also aiming to
prevent future
PPE supply
shortages by
establishing
long-term face
mask production
for the US
market. The site
manufactures
surgical masks
and community
masks and is
pursuing NIOSH
N95 respirator
certification.
Lydall Urges
Consumers to Buy
ASTM-Certified
and Buy
American-Made
ASTM
International
has introduced
new
certification
standards for
General Barrier
Face Coverings,
giving the
general public
greater
awareness and
control over the
level of
protection
provided by
cloth face
masks. ASTM
Level 1 Face
Coverings block
at least 20% of
particles 0.3
micron and
larger
(including
bacteria and
many viruses),
while ASTM Level
2 Face Coverings
block at least
50% of these
particles.
In response to
these new
standards, Sara
Greenstein,
President & CEO
of Lydall,
Inc.,
provided the
following
statement:
“Lydall and our
team of
filtration and
materials
science experts
were honored to
work alongside
ASTM
International,
the CDC, INDA
and our fellow
industry
colleagues in
this joint
effort to better
educate
consumers about
the level of
protection face
masks provide
against threats
like COVID-19.
Since the onset
of the pandemic,
manufacturers
all over the
world have
stepped up to
accelerate
production of
personal
protective
equipment and
the materials
they require but
demand still
heavily
outweighs
supply. For that
reason, it
remains
critically
important that
we right-size
our face masks
for our level of
risk – and in
order to do
that, we must
understand
exactly how much
protection masks
offer. N95
respirators and
surgical masks
that contain
fine fiber
meltblown
filtration media
remain the gold
standard, but
these new
standards
provide
much-needed
guidance for a
broader range of
more widely
available masks.
The message is
clear: by buying
ASTM-certified
and buying
American-made,
consumers can
have clarity and
confidence in
the level of
defense their
masks deliver
against the
virus.”
Handanhy
Supplies Melt
Blown Media and
Masks Worldwide
Handanhy is an
integrated
enterprise which
includes the
research and
development,
production and
sale of
filtration
media,
respirators and
coveralls. The
company was
founded in 1988
Heng Yong
Filtration
Material Factory
was built in
Yongnian County
starting with
only two
PP filtration
machines.
Today the
company has
broad portfolio
consisting of
·
Air Filtration
Media
·
Mask &
Respirator
·
Protective
Clothing
The company has
800 employees
and 100,000 m2
of factory
space.
More than 80% of
products are
exported,
Customers are
spread all over
the globe:
Britain, France,
Sweden, Holland,
Belgium, Greece,
America, Canada,
Russia, Iran,
Japan, Ireland,
France, Germany,
Singapore,
Malaysia,
Thailand, Chile,
Mexico,
Australia,
Spain, Norway,
Netherlands,
Estonia and
others.
The company has
been certified
by ISO
9001:2008 and ISO
14001:2004.
The masks have
been validated
for EN
149:2001+A1:2009 (notified
by BSI-CE 0086) and AS/NZS
1716:2003
(certified by
SAI Global,
Australia).
The company
specializes in
various types of
filter media,
such as
polypropylene
filter media,
polyester filter
media and
composite filter
media. Process
technologies
include
melt-blown,
needle-punched
and laminated
composites
(adhesive,
ultrasonic,
etc.). The
products are
widely applied
in industrial
air filtration;
vacuum cleaner
filtration;
vehicle cabin
filtration; and
protective
respirators and
clothing.
The company also
makes surgical
masks and
respirators.
Disposable
Particulate
Respirators
approved to EN149:2001+A1:2009 are
designed to
reduce the
wearer’s
exposure to
airborne
particles.
Mann + Hummel
Room Air
Purifier
Provides
Effective
Protection From
Viruses in
Classrooms
M+H introduced a
new room air
purifier in
December. The
HEPA H14 filters
(individually
tested according
to ISO 29463 &
EN 1822)
installed in the
two new mobile
and stationary
high-performance
room air
purifiers,
OurAir TK 850
and OurAir SQ
2500, are
capable of
reliably
filtering more
than 99.995
percent of
viruses,
bacteria and
micro-organisms
from the air. As
such, they help
to minimize the
risk of
infection in
internal spaces.
The devices are
modern in their
design, and
thanks to the
innovative ePTFE
medium, they
operate at high
performance with
particularly low
noise levels.
This allows
infectious virus
particles such
as SARS-CoV-2,
which can attach
themselves to
aerosols, to be
almost
completely
filtered out.
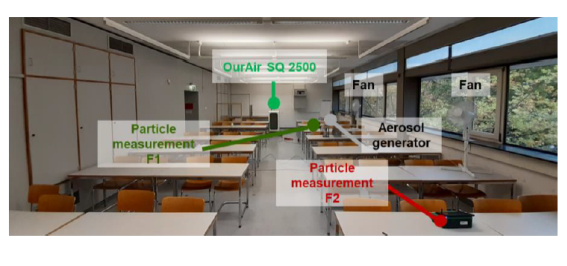
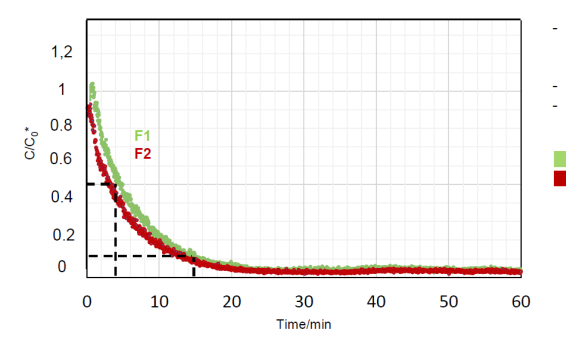
Investigation
of the reduction
of the aerosol
level by using
the OurAir SQ
2500
was conducted in
a class room and
measurement
taken at two
locations.
Significant
equal reduction
of the aerosol
levels at both
measuring points
F1 and F2
occurred
after switching
on the indoor
air purifier
(The particle
concentration at
F2 decreases
slightly faster
than at F1) 50%
after 4 min. /
90% after 15
min. Operation
below the
designed filter
rate (~ 5 h-1 )
results in a
slower
reduction.
Mann + Hummel
also supplies
masks. Students
can be protected
by masks in
addition to the
room air
purifier in the
classroom.
In the
lunchroom, the
room air
purifier with
more substantial
air flow and
laminar
direction can
compensate for
mask removal to
eat.
The company also
has the qLair
division which
can provide the
monitoring and
controls to
insure a safe
bubble within a
school or
restaurant.
Capture Virus
and Dust at the
Source - Brake
Filter Example
Mann + Hummel is
taking a
holistic
approach to
reducing indoor
and outdoor air
pollution.
The general
principle is to
eliminate the
pollution at the
source or as
close to the
source as
possible.
A good example
of innovative
source control
is the brake
particle filter
described below.
This filter
captures dust
from braking
with a minimal
amount of air.
Filter cubes
located at
traffic
intersections
capture
pollutants
relatively close
to the
source. The M+ H
air purifiers
can be used to
capture viruses
in local areas
of either high
virus
concentration or
where masks are
impractical e.g.
restaurants.
M+H has acquired
a mask company
(Purar). In the
holistic
approach people
have to be
viewed as a
major source.
The fact that
inefficient
cloth masks are
tolerated when
70 times more
virus can be
captured by a
tight fitting
efficient mask
is like an
industrial dust
collection
system
where there is
no dust hood for
the major
source. So in
all cases it
pays to capture
contaminants at
the source if
possible. Here
are the details
on the brake
filter
Fine dust is a
danger to human
health. However,
the fine dust is
not only caused
by exhaust
emissions: each
braking action
releases
particles. The
new brake dust
particle filter
from MANN+HUMMEL
will retain
these emissions
directly at the
source.
As the level of
air pollution
worldwide is on
the rise, the
awareness of
risks and
consequences to
human health is
also increasing.
Many cities
plagued by
contaminated air
are constantly
discussing the
causes and
possible
solutions.
Particles with a
diameter of less
than
10 micrometers
(PM10)
are particularly
dangerous
because they can
penetrate
pulmonary
alveoli. Smaller
particles than
that can even
directly enter
the blood and
cause heart
disease and
respiratory
illnesses.
The focus of
current
legislation is
concentrated on
the exhaust
emissions of the
combustion
engine. Other
sources of fine
dust such as the
fine dust
generated by
brake abrasion
have up to now
been disregarded
to a great
extent. In
Germany
approximately
10,000 tons of
brake dust
particles are
created every
year.
Measurements on
test benches
show that more
than 90% of this
consists of
particles with a
diameter smaller
than 0.55
micrometers. The
fine dust
pollution is
especially high
at busy road
junctions
because at such
locations many
braking actions
are required.
But longer
visits to
underground
railway stations
can also be
harmful to
health.
Measurements in
the London
underground
system, for
example, have
registered air
pollution with
inhalable
particles in the
range of 500 to
1,120 micrograms
per cubic meter.
The daily limit
specified by the
European Union
of 50 micrograms
per cubic meter
is therefore
greatly
exceeded. But it
is not only
vehicles with a
combustion
engine or trains
which create the
fine dust. In
electric and
fuel cell cars
with no exhaust
emissions and
stationary
engines braking
actions also
take place which
result in the
generation of
dangerous brake
dust.
MANN+HUMMEL has
developed a new
solution for all
of these
applications in
the form of the
brake dust
particle filter.
Located close to
the brake and
therefore the
source, the
filter retains
the particles
which are the
result of
mechanical
abrasion on the
brake. The
filter consists
of a robust
housing which is
positioned
directly in
continuation to
the brake
caliper and
captures
particles
created in the
braking process.
The new
development can
be fitted to any
existing
installation
space and can be
adapted to
different brake
sizes and
concepts.
One of the first
field tests for
the brake dust
particle filter
was made in the
‘fine dust
eater’ vehicles
from
MANN+HUMMEL.
These vehicles
have been on the
road for a
number of
months. Their
mission is to
retain more fine
dust than the
vehicles
generate
themselves. To
make this a
success, the
‘fine dust
eater’ vehicles
are equipped
with three
different types
of filter
systems: a fine
dust particle
filter, a new
type of NO2 fine
dust combi
filter for the
cabin and a
brake dust
particle filter.
For MANN+HUMMEL
this is a
further step
towards
implementation
of the vision
zero emissions.
It will Take
Years to
Vanquish
COVID-19
It will take
years, not
months, to gain
the upper hand
in the
coronavirus
pandemic — and
it will require
thinking well
beyond our
borders, says
Dr. Larry
Brilliant.
The 76-year-old
Bay Area
epidemiologist,
who has worked
to eradicate
smallpox, polio
and co-founded
the Seva
Foundation in
Berkeley to
combat
blindness, has a
unique
perspective on
the subject.
For years,
Brilliant warned
of a pandemic on
the scale of the
one we are
living through.
He even served
as the senior
technical
adviser on the
2011 motion
picture
“Contagion,”
filmed partly at
the San
Francisco 49ers
old stadium,
Candlestick Park
— during which
time he
presciently
predicted that
epidemiologists
would become
rock stars and
stadiums would
become mass
vaccination
sites.
As the CEO of
Pandefense
Advisory,
Brilliant has
assisted in the
global response
to the
coronavirus
crisis over the
past year. He
has a broader
view of the
pandemic than
most public
health leaders
and scientists,
having invested
his life’s work
in anticipation
of this very
moment.
He is encouraged
by dropping case
counts, the
rapid
development and
rollout of
innovative new
vaccines, the
new
science-driven
leadership in
the White House.
But Brilliant is
not here to
reassure anyone
that this will
all be over
soon.
“I think there’s
a lot of good
happening,” he
said, speaking
to The
Chronicle by
phone from his
home in Mill
Valley in a rare
interview
Friday. “I would
prefer to think
that things will
get better, but
the scientist in
me worries we’re
too perilously
close to things
going the other
way.”
Brilliant, who
moved to San
Francisco in
1967 from his
native Detroit,
clearly
remembers how he
felt the moment
he realized, in
January 2020,
that his
predictions were
about to come
true. He was in
a meeting with
top
epidemiologists
and public
health policy
experts from
around the world
at the Flood
Building in San
Francisco when
an alarming call
came in. The
Thai Health
Ministry had
identified a
case of the
novel
coronavirus at
one of its
airports.
“Those of us who
had been through
MERS and SARS,
we looked at
each other and
said, ‘This is
going to really
bad,’” Brilliant
said. “We knew
then that this
was going to
happen.”
As the first
leader of
Google’s
philanthropy
division,
Brilliant is
connected with
tech leaders and
public health
officials
worldwide. But
in the former
administration,
he found his
efforts stymied,
and he was
forced to watch
from the
sidelines as
every
opportunity to
get the pandemic
under control
was ignored,
dismissed and
disparaged.
He feels renewed
optimism with
President Biden
in charge,
especially with
a new federal
COVID-19
response team
that includes
Andy Slavitt,
the former
acting chief of
Medicare and
Medicaid under
the Obama
administration;
Vivek Murthy,
the former U.S.
surgeon general;
and David
Kessler, the
former
commissioner of
the Food and
Drug
Administration.
“I think it’s
going to be a
very different
attitude,”
Brilliant said,
praising Biden’s
swift action in
securing enough
vaccine to
deliver doses to
every American
by the end of
the summer.
“We’ve got
enough vaccine
on order to
vaccinate every
man, woman and
child in the
United States,”
he said. “We
also have enough
vaccine to help
the rest of the
world, which is
important.”
He cautioned,
however, that it
will take a
global effort to
truly make an
impact,
especially to
head off
increasingly
aggressive
variants of the
virus that could
elude testing
and vaccines.
Even if the
United States
gets most people
vaccinated, it
won’t be enough,
he said: “Until
everybody in the
world is safe,
no one is safe.”
The virus does
not recognize
borders, and
variants such as
those discovered
in the U.K.,
Brazil and South
Africa will
continue to
emerge until
everyone is
vaccinated, he
predicted.
Brilliant, a
longtime
Grateful Dead
doctor, laments
that half the
country still
believes in an
America First
policy, and that
kind of
nationalism will
create an
“uphill battle”
in the quest to
wipe out the
pandemic.
While he is
confident that
the vaccines in
production now
will be suitable
to combat the
variants in
circulation,
that picture
could rapidly
change.
“Today’s
vaccines are
good enough to
stop today’s
variants,”
Brilliant said.
“The problem is
tomorrow’s
variants. We’re
just on the
cusp.”
He thinks that
the global
picture of the
pandemic is much
more dire than
the numbers
show. There have
been more than
100 million
cases of
COVID-19
reported since
the start of the
pandemic,
according to
data collected
by Johns Hopkins
University.
Brilliant
predicts the
actual number of
cases will top a
billion,
“without
question.”
As government
leaders rush to
reopen the
economy and
schools while
the pandemic
continues to
rage Brilliant
urges caution.
“People are
beginning to
think, ‘Well, I
can go hug my
granddaughter,”
he said, noting
he rarely
ventures out of
the home he
shares with his
wife, Girija
Brilliant. “The
psychological
letdowns people
will have are
going to be
tremendous.”
He said the
three surges we
experienced last
year may not
have been surges
at all but a
portent of what
is to come
globally.
“There’s a
nonzero
probability that
we’ll look back
and all those
three bumps will
all together
just be the
first wave, and
what’s coming
next could be
worse,” he said.
“I think the
future is really
complicated.”
